A Blueprint for the Future
A unique collaboration between industry and regulators is aiming to understand what possibilities may exist to harmonize or consolidate PD-1/PD-L1 assays
At a Glance
- Immunotherapy – and in particular immune checkpoint targeting –is a rapidly growing area of cancer treatment
- Because of this rapid growth, multiple assays for pathways like the PD-1/PD-L1 checkpoint are under simultaneous development, but are not interchangeable
- Members of the pathology community have expressed concerns over the negative impact that recommending a single therapeutic, based on a single companion diagnostic, might have on patient care, laboratory costs, and workload
- To address concerns, the Blueprint Project, which is an unusual collaboration between industry and regulators, was set up to characterize and compare four PD-1/PD-L1 companion diagnostics
There’s no doubt in people’s minds that immunotherapy is an up-and-coming area of cancer treatment. In fact, some even think certain pathways – like the PD-1/PD-L1 immune checkpoint – are receiving an unfair share of the collective attention. Whether or not that’s true, one thing is for sure: there are a lot of PD-L1 expression assays currently under development. Surely, though, that’s a good thing – the more research, the better? Not necessarily – and in this case, with two existing PD-L1 therapies and at least four distinct companion assays in the pipeline, the US Food and Drug Administration (FDA) has expressed concern over the potential for future market confusion and, as a result, patient safety issues. But what can be done to avoid this from happening? Six companies (four pharmaceutical; two diagnostic) currently competing to bring their assays to market have taken an unusual step – they’re working together on an initiative known as the Blueprint Project to compare and characterize their tests.
Why do it?
In February of 2015, the six Blueprint sponsors (Bristol-Myers Squibb, Merck & Co., AstraZeneca, Roche, Dako/ Agilent and Ventana/Roche Tissue Diagnostics) met with the FDA and the American Association for Cancer Research (AACR) to discuss the concerns. After a public workshop to examine the issues, the sponsors put forward a proposal for the Blueprint PD-L1 Assay Comparison Project.
Why was it so important to establish this kind of comparison? It was clear from clinical trials that each assay had unique scoring guidelines and cutoffs used to identify responding patient populations – a fact that further fueled FDA concerns over the potential for patient safety issues if the assays were used to identify patients for cross-matched therapies. It’s important to be able to transpose results from one assay onto another – but at the time the Blueprint Project was initiated, no analytical comparison studies existed between any of the PD-L1 tests going through drug- diagnostic co-development. Obviously, companies generally don’t collaborate on pre-market evaluations like this one. The PD-L1 situation is unique, though; never before have so many companion diagnostic combinations been developed for the same biomarker at the same time. The Blueprint sponsors agreed that the situation – and the concerns that went along with it – required an uncommon approach.
What is the Blueprint Project?
The project involves a number of parties, each playing a unique part. The FDA is the overall observer and monitors the process; the AACR facilitates conversations and provides project updates; the pharma and in vitro diagnostics companies provide support in the form of resources and technical expertise; and the International Society for the Study of Lung Cancer acts as a neutral observer and provider of pathology expertise.
To begin, the FDA and AACR identified the sponsors who were most advanced in the clinical trials process with “Investigational Use Only” PD- L1 assays and drugs. During initial meetings, the scope of the project was refined to include only non-small cell lung cancer (NSCLC), as those tended to be the most advanced trials. Other scope refinements meant that only assays that would eventually be submitted for FDA review and approval via the Premarket Approval (PMA) process were included – excluding lab-developed tests. Ultimately, assays for four agents – nivolumab, pembrolizumab, durvalumab and atezolizumab – were selected.
In Phase One, teams from Ventana and Agilent selected procured NSCLC specimens to demonstrate the full dynamic range of each assay, then stained the slides using their own solutions with each of the four PD-L1 IHC assays. The analysis was fairly detailed. First, with no prior training or pre-alignment, two pathologists from Ventana and one from Agilent (experts in interpreting their respective assays) independently evaluated 156 IHC slides (39 cases for each of the four assays) for percentage of tumor cells and percentage of immune cell expression of PD-L1. Each expert pathologist also independently evaluated each case using only the clinical algorithm associated with their own assay of interest; for example, the 22C3 expert pathologist read all slides using the 22C3 selected cutoff. Then, the validated clinical diagnostic PD-L1 determinations for each assay were compared on a cohort of cases to demonstrate how hypothetical treatment decisions might be made for the four therapeutics. And, finally, the agreement rates of various combinations of assays and cutoffs were examined.
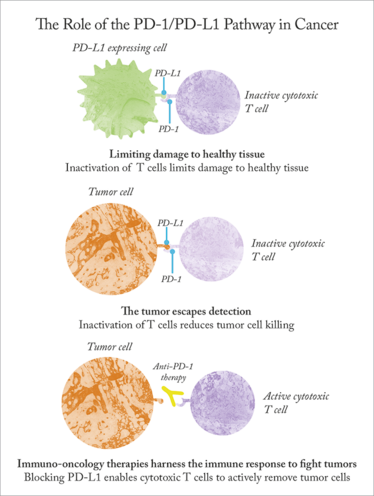
Phase One of the Blueprint Project was originally defined as a premarket study. One condition of its existence was that it would not delay pivotal studies or patient access to critical new therapies. In March of 2015, no PD-L1 assays or drugs had been approved – but during the course of the Phase One study, Agilent had two PD-L1 tests receive three premarket approvals (PMA), and several drug registrations in different indications occurred. Patient selection criteria were changing, both in the market and in clinical trials based on new data. Additional drug and diagnostic filings were in process. All of these activities took priority in, but also created challenges for the Blueprint Project, as all of these dynamics created a “moving target” for analytical assay comparison. That made it more complicated to reach agreement on the data analysis approach and factors to be compared. Eventually, however, initial results were delivered showing that three of the four assays evaluated demonstrate similar PD-L1 expression in tumor cells, while all four were shown to be variable (with lower reproducibility) in immune cells.
Onto the next phase
The primary purpose for Phase Two of the project is to validate the initial findings of Phase One using a much larger set of samples that better reflect “real-world” conditions. Phase Two will also likely assess variability between test sites and observers to better understand the magnitude of those variables and their impact on testing robustness. The International Association for the Study of Lung Cancer (IASLC), which was originally selected to participate in Blueprint as a third party observer, is well-positioned to plan and execute the Phase Two study with support from the project’s sponsors. Together, they’re working out the study scope and design, but at the moment, that remains a work in progress.
“Given its significance in many cancers, I envision that PD-L1 testing will continue to play a major role in identifying patients who may benefit from immune checkpoint inhibitors.The hope is that testing can be simplified in the future as we learn more about PD-L1 expression in cancer.” – Henrik Winther
“Pathologists should be aware that, when they use PD-L1 IHC tests to inform therapy decisions, it’s important to follow the validated assays’ intended use. Despite the analytical similarities between some assays, the conclusion so far is that they are not interchangeable when selecting patients for PD-1 or PD-L1 therapies. Each test is developed and validated independently to identify corresponding populations for individual drugs, and they should only be used for their specific drug. As long as the tests are used appropriately, they could lead to significant improvements in treatments not just for NSCLC, but eventually for other diseases as well.” – Hans Christian Pedersen
PD-L1 immunohistochemistry isn’t limited to NSCLC, though; it’s being explored in many indications beyond lung cancer, and Blueprint serves as proof-of- concept for this kind of collaboration within industry. It will definitely be valuable for companies to continue working together to show analytical correlation between tests and data on assay performance in NSCLC. The Blueprint Project and its successors will likely influence how future indications for PD-L1 IHC are designed, both in terms of assay development and showing clinical utility.
Entry into the field of complementary diagnostics adds a new category for approval, the influence of which is yet to be determined. New testing technologies and platforms will require new paradigms to determine clinical utility and gain regulatory approval, but no matter what happens next, the future of companion and complementary diagnostics promises to be a dynamic one.
Words to the wise
Many members of the laboratory medicine community have expressed concerns over the negative impact that recommending a single therapeutic, based on a single companion diagnostic, might have on patient care, laboratory costs, and workload. These concerns were the main drivers for the initial public workshop in 2015, and for the project that ultimately arose. The analytical performance comparison of PD-L1 assays is the first step to understanding what possibilities may exist to harmonize or consolidate – but this will be an ongoing discussion for some time between pharmaceutical companies, diagnostic companies and regulatory agencies.
Henrik Winther is Vice President and General Manager of Companion Diagnostics in Agilent’s Diagnostics and Genomics Division.
Hans Christian Pedersen is Head of Companion Diagnostics and Immunohistochemistry Reagents for Agilent Pathology Solutions.
Henrik Winther is Vice President and General Manager of Companion Diagnostics in Agilent’s Diagnostics and Genomics Division.
Hans Christian Pedersen is Head of Companion Diagnostics and Immunohistochemistry Reagents for Agilent Pathology Solutions.