
Liquid biopsy comprehensive genomic profiling (CGP) is emerging as a powerful complement to tissue-based testing, offering earlier molecular insights, faster turnaround times, and expanded access to targeted therapies – particularly in lung and breast cancer.
In a recent webinar supported by Illumina, Pilar Ramos, Senior Manager at Medical Affairs Europe, Illumina, and Rachel Dodds, Principal Scientist at All Wales Medical Genomics Service (AWMGS), outlined both the technical foundations of circulating tumor DNA (ctDNA) analysis and its real-world implementation in routine clinical pathways. The session highlighted how liquid biopsy can address persistent limitations in tissue testing, while also underscoring the operational and interpretive challenges laboratories must navigate as adoption increases.
Learning objectives
Understand the fundamentals of liquid biopsy comprehensive genomic profiling (CGP) and how it complements tissue testing
Learn where liquid biopsy provides clinical value, especially in lung and breast cancer pathways
See how earlier molecular insights can support faster, more informed treatment decision making in routine practice
Gain insights from real world implementation in Wales, including workflow integration and ESR1 testing in breast cancer
Recognize common operational and clinical challenges during liquid biopsy adoption – and strategies to overcome them
Addressing gaps in tissue-based genomic profiling
Despite advances in precision oncology, access to comprehensive genomic data remains inconsistent. “Up to 30 percent of patients don’t fully benefit from precision medicine because there isn’t enough tumor tissue available for next generation sequencing profiling,” noted Ramos.
This limitation is particularly pronounced in cancers such as lung cancer, where biopsy samples may be small, depleted by prior testing, or difficult to obtain. Incomplete molecular profiling can result in missed opportunities for targeted therapy.
Liquid biopsy offers a non-invasive alternative by analyzing ctDNA shed from tumors into the bloodstream. Although ctDNA typically represents less than one percent of total cell-free DNA, it can provide clinically actionable information across the cancer continuum – from diagnosis and treatment selection to monitoring and detection of resistance.
Ramos emphasized that liquid biopsy should not replace tissue testing, but complement it, adding, “While tissue biopsy remains the gold standard for biomarker testing, it has limitations. Liquid biopsy on the other hand is less invasive and can better reflect tumor heterogeneity.”
Capturing tumor heterogeneity and resistance
One of the key advantages of ctDNA analysis is its ability to capture tumor heterogeneity across primary and metastatic sites. Unlike a single tissue biopsy, which reflects one location at one time point, liquid biopsy aggregates DNA fragments from multiple tumor clones.
This broader view can be critical in identifying resistance mechanisms. In one case presented by Ramos, ctDNA profiling detected multiple resistance mutations that were not captured in any single tissue biopsy, demonstrating its value in guiding treatment decisions (Figure 1).
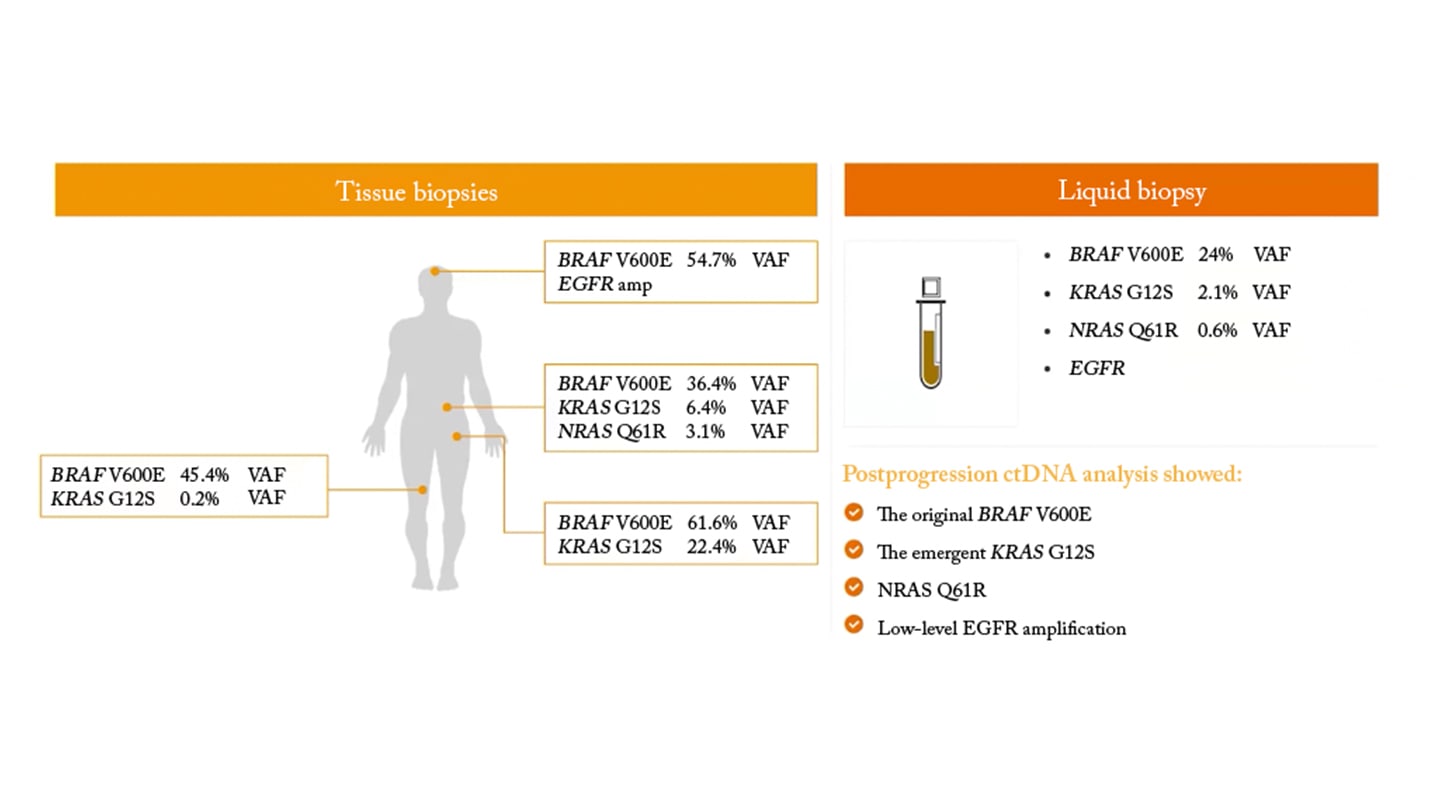
However, this approach introduces analytical challenges. Clonal hematopoiesis – age-related mutations in blood cells – can confound results, requiring advanced bioinformatics and matched normal sequencing to distinguish true tumor variants. Additionally, ctDNA levels vary by tumor type, stage, and timing of sampling, meaning not all patients are equally suitable candidates.
“These dynamics are critical because they help us choose the right moment to collect samples and ensure we get the most informative and reliable results,” Ramos explained.
Integrating liquid biopsy into lung cancer pathways
Dodds provided a detailed overview of the “quickDNA” initiative in Wales, which integrated liquid biopsy CGP into the lung cancer diagnostic pathway (Figure 2).
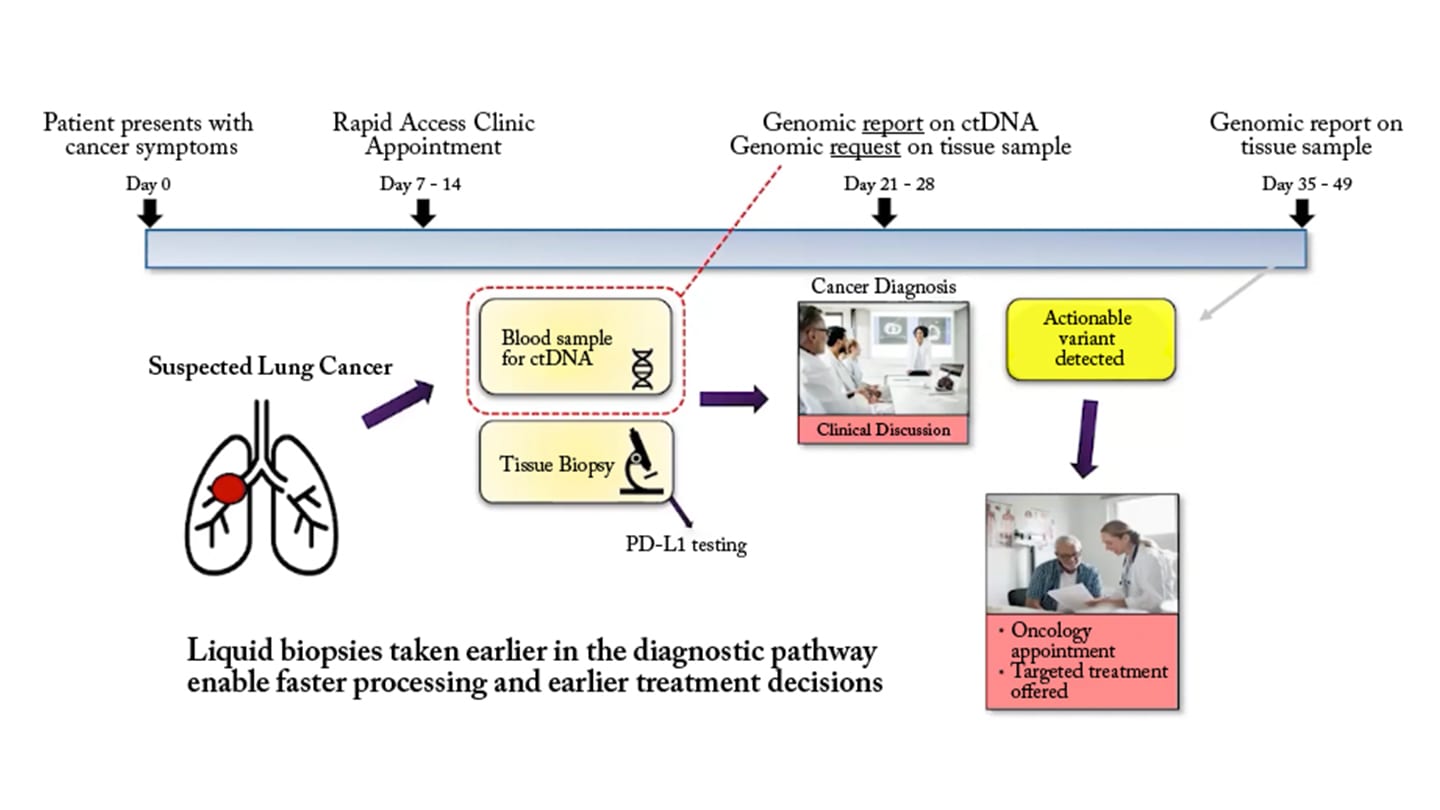
Lung cancer represents a high-priority target for innovation. It is the leading cause of cancer-related death in Wales, and patients often experience delays in accessing genomic testing due to the traditional sequence of diagnostic steps.
“In the traditional pathway, genomic testing is requested later, which is not ideal when that result can guide the patient's treatment,” Dodds said.
By introducing ctDNA testing earlier – at the point of referral to a rapid access clinic – the quickDNA study enabled genomic analysis to run in parallel with imaging and pathology. This approach significantly shortened the time to actionable results.
The impact was notable. Median turnaround time for ctDNA reports was 14 days, compared with 26 days for tissue testing. More importantly, time to treatment allocation was reduced from 98 days with tissue testing to 45 days with liquid biopsy in the study cohort.
“Our preliminary study of 67 patients demonstrated a clear benefit to enable access earlier to these targeted therapies,” Dodds said.
Expanding access to targeted therapies
Liquid biopsy also increased the number of patients eligible for targeted treatment. In the feasibility cohort, actionable variants were identified in 24 patients via ctDNA compared with 18 via tissue testing. Notably, some patients lacked adequate tissue samples entirely, highlighting the complementary role of blood-based testing.
Case studies illustrated the clinical impact (Figure 3). In one patient with an ALK rearrangement, ctDNA results were available 50 days earlier than tissue results, enabling rapid initiation of targeted therapy. In another case, where repeated tissue biopsies failed, ctDNA testing provided the only actionable result – leading to treatment and significant clinical improvement.
“Without the ctDNA result, this patient would not have received that targeted therapy,” Dodds noted.

Real-world implementation in breast cancer
Beyond lung cancer, the webinar highlighted the use of liquid biopsy in breast cancer, particularly for ESR1 mutation testing. ESR1 mutations are associated with resistance to aromatase inhibitors but may predict response to newer therapies such as elacestrant.
AWMGS implemented ctDNA-based ESR1 testing in early 2025, leveraging existing CGP infrastructure. Over six months, 153 samples were analyzed, with actionable variants detected in 46 percent of patients – consistent with published data.
Importantly, the service achieved rapid turnaround times, averaging 12.5 days, and demonstrated the ability to detect low-frequency variants, including those present in subclonal populations.
“Tumors do contain different populations of cells and ctDNA is better able to avoid sampling bias and capture the full genomic profile,” Dodds explained.
The ability to detect multiple ESR1 variants within a single sample further supports the use of liquid biopsy in monitoring tumor evolution and guiding therapy in metastatic disease.
Workflow and operational considerations
Successful implementation of liquid biopsy requires careful attention to pre-analytical and analytical workflows. Sample handling is particularly critical. Blood must be collected in specialized preservative tubes and processed promptly to prevent white blood cell lysis, which can dilute ctDNA and compromise sensitivity. Quality control measures, such as fragment size analysis, are essential to ensure accurate results.
“Pre-processing and sample quality is very important; you need to make sure that you're not looking at a sample that has been contaminated,” Dodds said.
Analytically, ctDNA testing demands higher sensitivity and different interpretation frameworks compared with formalin-fixed, paraffin-embedded (FFPE) tissue. Laboratories must also address challenges such as low tumor fraction, variant calling at low allele frequencies, and discordance between tissue and liquid biopsy results.
Negative results, in particular, require cautious interpretation. As Dodds noted, absence of detectable variants does not necessarily indicate absence of disease, especially in cases with low ctDNA shedding.
Evolving role in clinical practice
Guidelines are increasingly recognizing the role of liquid biopsy. Both ESMO and NCCN now support ctDNA testing in specific clinical contexts, particularly when tissue is unavailable or rapid decision making is required.
Looking ahead, both speakers emphasized that liquid biopsy will continue to expand across indications, tumor types, and sample sources. Ongoing developments include broader gene panels, improved tumor fraction estimation, and integration with other modalities such as methylation and fragmentomics.
“I think we’re just starting to see the value of this approach in routine testing,” Ramos said.
This evolution presents both opportunities and challenges. Liquid biopsy has the potential to streamline workflows, improve patient access to precision therapies, and provide deeper insights into tumor biology – but it also requires investment in infrastructure, training, and standardization.
Conclusion
The integration of liquid biopsy CGP into clinical pathways represents a significant step forward in precision oncology. By complementing tissue testing, enabling earlier intervention, and expanding access to actionable genomic data, ctDNA analysis is reshaping diagnostic and treatment paradigms in lung and breast cancer.
Real-world data from Wales demonstrate that these benefits are achievable in routine practice, provided that laboratories address key operational and analytical challenges.
As adoption grows, collaboration between clinicians, pathologists, and laboratory scientists will be essential to fully realize the potential of liquid biopsy and ensure its effective implementation across health systems.





