
Evaluating rapid assays for the detection of somatic mutations in tissue and liquid biopsies
Digital PCR (dPCR) has emerged as a versatile tool across multiple oncologic applications. In addition to rapid screening for actionable mutations, the technology can be used to monitor minimal residual disease, detect low-frequency molecular alterations, identify mutations in histologically normal tissue, and support liquid biopsy-based approaches.
In a webinar sponsored by QIAGEN, Dr Valentina De Pascale, of the IRCCS Regina Elena National Cancer Institute in Italy, presented the results of a validation study of dPCR assays to detect TERT and KRAS mutations in patient tissue samples. The dPCR assays demonstrated 100 percent concordance with reference methods while offering a faster workflow and minimal hands-on time.
Finally, Lise Grewis, CEO of ID Solutions, presented her future vision for rapid testing of actionable mutations in oncology.
Learning objectives
Compare the performance and concordance of rapid dPCR assays with next-generation sequencing (NGS) for detecting somatic mutations in tumor tissue and liquid biopsy samples.
Evaluate the benefits of rapid dPCR assays for use in routine mutation screening.
Explore the promise of dPCR liquid biopsy testing in the future of oncology diagnostics and disease monitoring.
The presented workflow is only valid for use within the health institution that developed and validated it, IRCCS Regina Elena National Cancer Institute (IRE). The work was performed under the regulatory requirements laid out in Article 5(5) of the In Vitro Diagnostic Medical Devices Regulation (IVDR 2017/746).
The ID SOLUTIONS IDNAPTEX dPCR Assays and QIAGEN QIAcuity dPCR systems described here are intended for non-clinical applications. These products are not intended for the diagnosis, prevention or treatment of a disease.
Rapid and reliable cancer mutation detection with dPCR

dPCR performance evaluation
The investigation was structured around three primary aims:
Evaluate the technical performance of rapid dPCR assays using formalin-fixed, paraffin-embedded (FFPE) tumor samples.
Compare dPCR assays with complementary molecular analysis methods to assess concordance and clinical validity.
Explore potential clinical and research use cases, assessing whether rapid dPCR-based testing could be feasibly integrated into routine diagnostic workflows.
In this experimental setting, the investigators employed the QIAcuity dPCR platform. Based on nanoplate technology, it physically partitions each sample into thousands of discrete reaction chambers. Each partition is analyzed independently, enabling absolute quantification of target sequences without the need for calibration curves.
“These characteristics allow for very high sensitivity and precise quantification,” Dr De Pascale explained, noting that such performance features have significantly expanded the role of dPCR in molecular diagnostics.
The investigators evaluated three IDNAPTEX multiplex dPCR assays, each targeting clinically actionable alterations relevant to solid tumor diagnostics.
TERT promoter mutations – the first assay was designed for the detection of the two most prevalent variants, C228T and C250T, which are frequently observed across multiple tumor types and represent high-value targets for rapid screening in FFPE tissue.
KRAS G12C – although less common than other KRAS substitutions in colorectal cancer (CRC), G12C has gained increasing clinical relevance with the development of mutation-specific targeted therapies, making its accurate and timely identification essential.
Comprehensive KRAS mutation panel – the third assay was designed to detect the most frequent alterations across exons 2, 3, and 4 of the KRAS gene. Collectively, mutations in these regions account for the majority of KRAS alterations observed in CRC and are also prevalent in other solid tumors, including non-small cell lung cancer.
TERT promoter dPCR analysis shows workflow efficiencies and practical advantages
The study adopted a simple workflow – comprising DNA extraction, test preparation, and loading into the dPCR system – which yielded results in a matter of hours (Figure 1). The validation phase involved reproducibility tests, establishing the limit of blank, and the method comparison test.

The investigators compared the ID Solutions dPCR kit with an in-house custom dPCR assay in the evaluation of TERT promoter mutations in 30 samples from patients with known TERT promoter mutations variants – 15 each with C228T and C250T mutations.
While manual laboratory hands-on time was comparable between the two approaches, several practical differences emerged that favored the commercial solution. First, the ID Solutions kit had immediate availability and came with a warranty and support as standard – an important consideration for laboratories seeking standardized assays suitable for routine research use.
Second, the mutation status of the two most common TERT promoter variants – C228T and C250T – could be assessed simultaneously in a single analysis. That represented notable time and cost savings compared with the custom assay, which required separate reactions for each mutation (Figure 2).
Importantly, the IDNAPTEX dPCR method demonstrated 100 percent sensitivity and specificity across all 30 samples, as well as complete concordance with the reference method (ddPCR).

KRAS dPCR analysis is quick and accurate, even with limited DNA
For KRAS mutation analysis, the investigators compared the ID Solutions dPCR assays with NGS, the current reference standard for comprehensive KRAS testing.
One of the most immediate differences was hands-on laboratory time. Preparing samples for dPCR required only a few minutes, whereas an NGS workflow involved several hours of manual processing, including library preparation and quality control steps (Figure 3).
Turnaround time was further impacted by downstream analysis. NGS requires a dedicated bioinformatics pipeline and data interpretation, extending the time to results. In contrast, dPCR analysis using the ID Solutions kit was described as faster and more straightforward, with mutation calls that were immediately interpretable.
“With dPCR, results are available within hours,” Dr De Pascale confirmed.
The assays also differed substantially in their input DNA requirements – a critical factor when working with small or degraded FFPE samples. Standard NGS workflows typically require approximately 20 ng of input DNA, whereas the dPCR assays evaluated in this study required less than 1 ng.
“While NGS remains essential for broad genomic profiling, dPCR presents a faster and more cost-effective solution for low-input KRAS mutation testing,” said Dr De Pascale.
Altogether, these findings highlight dPCR as a potentially robust approach for sensitive mutation detection and primary mutation screening, complementary to NGS.

Rapid and reliable ESR1 mutation detection with dPCR liquid biopsy testing
ESR1 mutations have emerged as key oncogenic drivers in ER-positive breast cancer. Their detection not only predicts response to endocrine therapies, but also informs the selection of targeted therapy with selective estrogen receptor degraders (SERDs).
Dr De Pascale’s final study was a preliminary analysis of the effectiveness of the ID Solutions IDNAPTEX ESR1 assay to monitor ESR1 mutated circulating tumor DNA in liquid biopsies over time.
The ESR1 assay was compared with custom assays in the analysis of twelve samples from two patients with confirmed ESR1-positive breast cancer at different time points (Figure 4). A further twelve samples of unknown mutational status from an ongoing multicenter study were also analyzed.
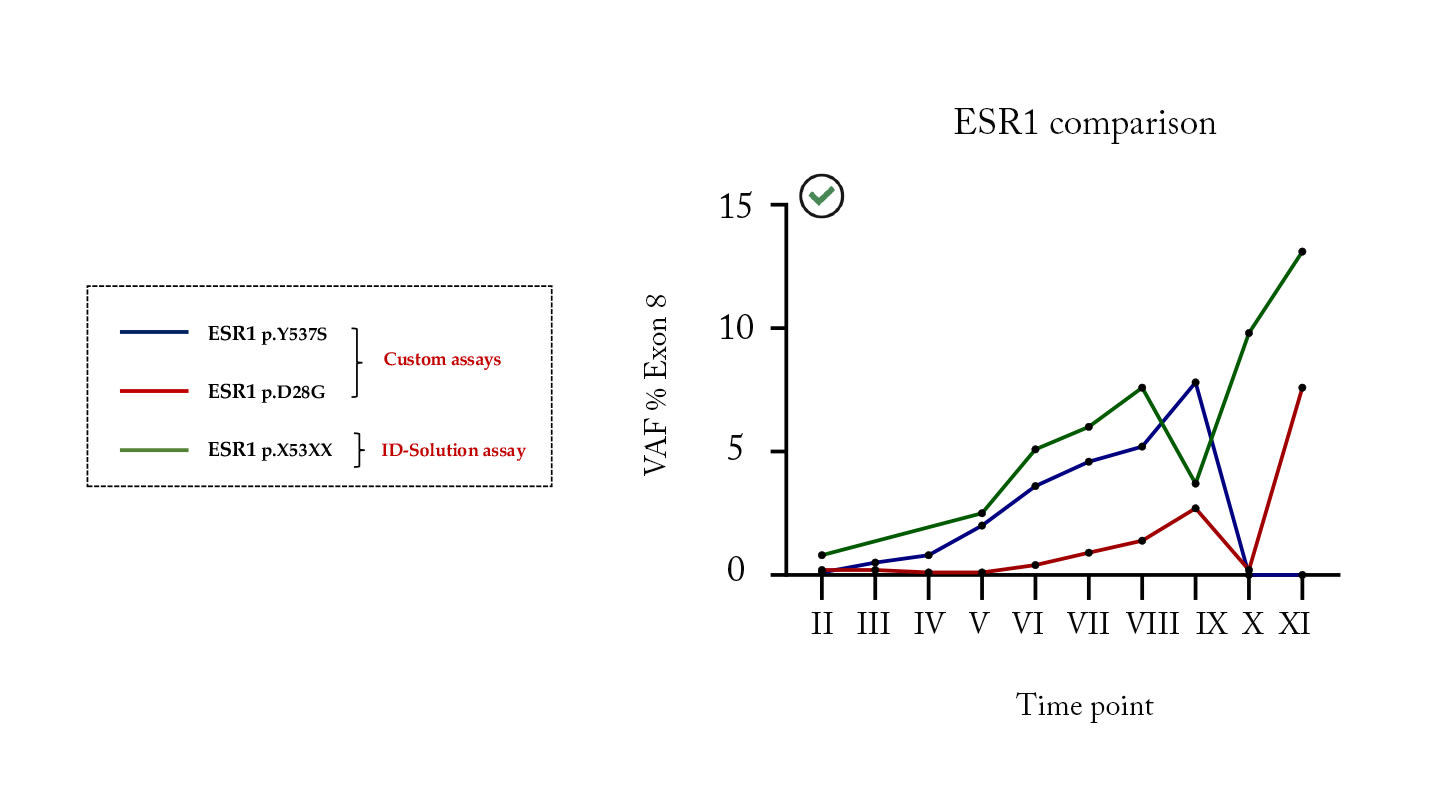
“In this limited number of samples, the results support the possibility of using dPCR for longitudinal monitoring of ESR1 in liquid biopsies,” Dr De Pascale reported.
dPCR for precision oncology
In summary, the studies demonstrated that the QIAcuity dPCR platform, when combined with ID Solutions assays, provides a flexible, robust, and highly reliable system for molecular analysis in oncology.
The evaluated assays enabled rapid and accurate detection of clinically actionable mutations in TERT, and KRAS, supporting their use as tools in the oncology research lab.
“They are also suitable for studies involving minimal residual disease monitoring and for detecting mutations at very low allele frequency,” Dr De Pascale stressed. “Meanwhile, the assays’ liquid biopsy applications represent an important future direction.”
“These capabilities highlight the growing potential of dPCR-based approaches to bridge the gap between advanced molecular research and routine clinical practice, positioning the technology as an increasingly powerful component of precision oncology,” Dr De Pascale concluded.
A powerful partnership for oncology research

Presenting ID Solutions
ID Solutions, based in France, has been pioneering liquid biopsy and dPCR tools in oncology since 2016. The company is dedicated to translating its patented technology for biomarker monitoring and early detection into comprehensive solutions, from sample to result. In 2025 QIAGEN partnered with ID Solutions to launch the IDNAPTEX multiplex dPCR assays for research use, designed for simultaneous detection of multiple tumor-associated DNA variants.
With liquid biopsy as the cornerstone of its approach, ID Solutions aims to reinvent oncology follow-up care with the emphasis on real-time monitoring of therapeutic response and early detection of disease progression and recurrence.
“Our vision is for proactive and personalized medicine, leading to greater peace of mind for patients,” Grewis explained.
Liquid biopsy as a therapeutic follow-up
ID Solutions’ ambition is to apply the ultra-sensitive detection powers of dPCR with liquid biopsy to generate accurate clinical data that can be used immediately.
“The body talks with the blood,” said Grewis. “In liquid biopsies we can detect mutation before relapse, up to six months before it shows on imaging.”
This testing approach will provide oncologists with the data they need for timely, tailored treatment decisions.
Putting technology at the service of life
“We all know what it means to be confronted with cancer,” Grewis said. “Each of us has been affected, directly or indirectly, by a loved one who is ill. This is the experience behind ID Solutions’ patient-centered approach.”
“The partnership of ID Solutions assays with QIAGEN’s QIAcuity dPCR systems provides a robust and complete research workflow that has the potential to help patients and oncologists, alike, everywhere in the world,” she concluded.





