A New Look at Leukemia and Lymphoma
Flow cytometric immunophenotyping can assist with the diagnosis of a range of lymphoid and myeloid neoplasms
Flow cytometry is an example of a rapidly evolving IVD technology that is migrating from the highly specialized laboratory to routine clinical testing in the core lab. It has been widely adopted for the assessment of leukemias and lymphomas and is part of the trend toward early detection and monitoring, bringing diagnostics closer to patients. 2017 marked a milestone for the technology when the US Food and Drug Administration (FDA) granted de novo authorization for the in vitro diagnostic use of the first pre-formulated antibody cocktail for leukemia and lymphoma immunophenotyping.
Flow cytometric immunophenotyping evaluates the presence and absence of specific antigens for each individual cell in the specimen. Interpretation of aberrant immunophenotypes requires skill in recognizing the significance of different patterns. As part of the test manufacturer’s available information, a unique case book has been created to aid in this complex pattern recognition. The following case is just one example.
Diagnosing B cell lymphoblastic leukemia/lymphoblastic lymphoma
A 33-year-old male presents with anemia, thrombocytopenia, and circulating atypical mononuclear cells. Using flow cytometric immunophenotyping, we are able to identify a phenotypically distinct population of cells with low light scatter properties that express CD10, CD19, CD34, and CD38. CD45 expression ranges from low density to very low density. Neither CD20 nor immunoglobulin light chain expression is noted.
Taken together, this is most consistent with a diagnosis of B lymphoblastic leukemia/lymphoma. Blast crisis in chronic myelogenous leukemia is also a diagnostic consideration. Correlation with clinical and laboratory data is recommended, and additional immunophenotyping may be warranted.
The dot plot in Figure 1 permits distinction of the usual populations including lymphocytes (Gate A, red), monocytes (Gate B, green), and granulocytes (Gate C, blue). Gate D (pink) is in the area typically occupied by myeloblasts, but may be used to highlight other populations. By applying different colors to the events captured by each gate, the various populations may be followed throughout the analysis (see Figure 2). Gates should be adjusted to conform to the naturally occurring separations among the populations but, where no separation is observed, users may make an estimate based on experience. The aberrant events in this sample fall largely within Gate D. Some of the events fall outside of Gate E, and Gates D and E may be adjusted.
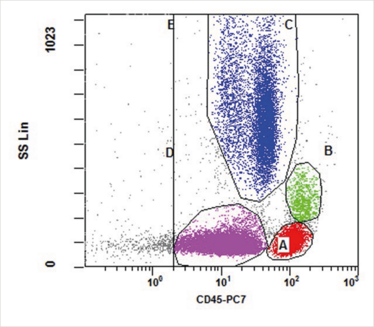
Figure 1. This CD45 vs. side scatter dot plot is ungated and shows all events collected. Gate E includes all CD45-positive events and may be used to set a stop count gate during acquisition to ensure that sufficient non-debris events are collected. Gate E may also be used to exclude CD45-negative debris from the analysis, but these events should not be ignored when analyzing a case, as some aberrant populations are CD45-negative.
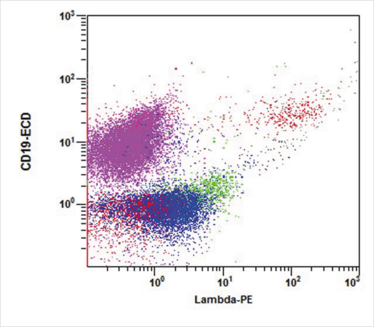
Figure 2. This lambda vs. CD19 dot plot is gated on E and shows all CD45-positive events. The CD19-positive events are split equally between lambda-positive and lambda-negative populations, consistent with polyclonal B lymphocytes. The aberrant population (pink) expresses CD19, but is negative for kappa immunoglobulin light chains.
The ClearLLab casebook includes 16 such illustrative clinical vignettes with characteristic findings typical of various lymphoid and myeloid neoplasms, as well as examples of patients with indications of an underlying neoplastic process, but in whom no immunophenotypic abnormality is identified. Users can also download the data to continue their diagnostic practice.
See http://info.beckmancoulter.com/casebook-usa for more information and to obtain a copy of the book and data.
The clinical flow team at Beckman Coulter Life Sciences are exhibiting at AACC 2018 at booth #3612. For more information on Beckman Coulter Life Sciences, see www.beckman.com/home